
Robert J. Thomas,
Scientific Solution
"When carrying out elemental determinations of complex samples with high concentrations of matrix components, such as clinical, pharmaceutical, geological, environmental or petrochemical matrices, it’s extremely important that the plasma spectrochemical instrumentation used is kept in good working order to maximize performance and productivity (1, 2). This is particularly the case with ICP-MS, one of the most widely used trace element techniques, which has unparalleled multielement detection capability. However, in the hands of a novice or inexperienced user, it can generate erratic and imprecise results, unless the instrument is maintained and cleaned on a regular basis. For that reason, it requires a certain level of operator competence and expertise to fully understand when a problem arises so preventative action can be taken."
ICP-MS Instrumental Components
The fundamental principle of inductively coupled plasma mass spectrometry (ICP-MS), which gives the technique its unequalled isotopic selectivity and sensitivity, also unfortunately contributes to some of its weaknesses. The fact that the sample “flows into” the spectrometer and is not “passed by it” at right angles, such as flame AA and radial inductively coupled plasma optical emission spectroscopy (ICP-OES), means that the potential for thermal problems, corrosion, chemical attack, blockage, matrix deposits, and drift is much higher than with the other atomic spectrometry (AS) techniques. However, being fully aware of this fact and carrying out regular inspection of instrumental components can reduce and sometimes eliminate many of these potential problem areas. There is no question that a laboratory which initiates a routine maintenance schedule stands a much better chance of having an instrument ready and available for analysis whenever it is needed, compared to a laboratory that basically ignores these issues and assumes the instrument will look after itself.
Let us remind ourselves of the ICP-MS instrumental components (shown in Figure 1) and in particular, those that require inspection and maintenance on a routine basis including:
- Nebulizer and spray chamber
- ICP torch
- MS interface region
- Ion optics
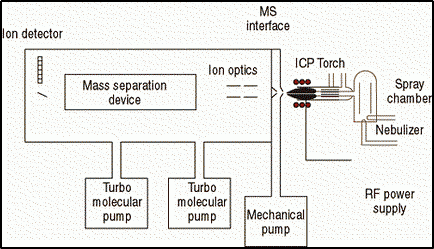
Figure 1: Instrumental components of an ICP-MS
As mentioned in my first blog, the majority of imprecision problems in ICP-MS arise due to the potential of sample matrix depositing itself either in the nebulizer tips, the sample injector of the torch and/or the interface cones and ion optics. In this month’s blog, we will focus on the mass spec interface region, the ion focusing system, and in particular how to minimize the amount of dissolved solids entering the mass spectrometer.
Interface Region
As the name suggests, the interface is the region of the ICP mass spectrometer where the plasma discharge at atmospheric pressure is “coupled” to the mass spectrometer at 10−6 torr by way of two interface cones—a sampler and skimmer. This coupling of a high-temperature ionization source such as an ICP to the metallic interface of the mass spectrometer imposes demands on this region of the instrument that are unique to this AS technique. When this is combined with matrix, solvent, and analyte ions together with particulates and neutral species being directed at high velocity at the interface cones, an extremely harsh environment is the result. The most common types of problems associated with the interface are blocking or corrosion of the sampler cone and, to a lesser extent, the skimmer cone. A schematic of the interface cones showing potential areas of blockage are shown in Figure 2.
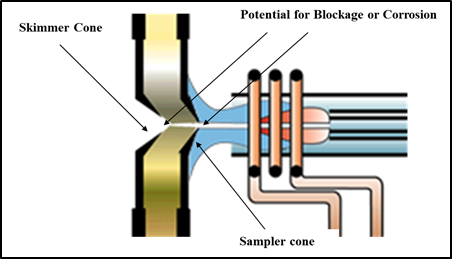
Figure 2: ICP-MS interface cones showing potential areas of blockage
A blockage is not always obvious, because often the buildup of material on the cone or corrosion around the orifice can take a long time to reveal itself. For that reason, the sampler and skimmer interface cones have to be inspected and cleaned on a regular basis. The frequency will often depend on the types of samples being analyzed and also the design of the ICP mass spectrometer. For example, it is well documented that a secondary discharge at the interface can prematurely discolor and degrade the sampler cone, especially when complex matrices are being analyzed or if the instrument is being used for high sample throughput.
Besides the cones, the metal interface housing itself is also exposed to the high-temperature plasma. Therefore, it needs to be cooled by a recirculating water system, usually containing some kind of antifreeze or corrosion inhibitor or by a continuous supply of mains water. Recirculating systems are probably more widely used because the temperature of the interface can be controlled much better. There is no real routine maintenance involved with the interface housing, except maybe to check the quality of the coolant from time to time, to make sure there is no corrosion of the interface cooling system. If for any reason the interface gets too hot, there are usually built-in safety interlocks that will turn the plasma off. Some useful hints to prolong the lifetime of the interface and cones include the following:
- Check that both sampler and skimmer cone are clean and free of sample deposits. The typical frequency is weekly, but will depend on sample type and workload.
- If necessary, remove and clean cones using the manufacturer’s recommendations. Typical approaches include immersion in a beaker of weak acid or detergent placed in a hot water or ultrasonic bath. Abrasion with fine wire wool or a coarse polishing compound has also been used.
- Never stick any wire into the orifice; it could do permanent damage.
- Nickel cones will degrade rapidly with harsh sample matrices. Use platinum cones for highly corrosive solutions and organic solvents.
- Periodically check cone orifice diameter and shape with a magnifying glass (10–20× magnification). An irregular-shaped orifice will affect instrument performance.
- Thoroughly dry cones before installing them back in the instrument because water/solvent could be pulled back into the mass spectrometer.
- Check coolant in recirculating system for signs of interface corrosion such as copper or aluminum salts (or predominant metal of interface).
Robert (Rob) Thomas has worked in the field of trace element analysis for over 40 years, including 24 years for an ICP-MS manufacturer and 17 years as a principal of his own consulting company, Scientific Solutions (www.scientificsolutions1.com) He has served on the American Chemical Society (ACS) Reagent Chemical Committee for the past 17 years as leader of the elemental impurities task force where he has worked closely with the United States Pharmacopeia (USP) to align heavy metal testing procedures in reagent chemicals with those of pharmaceutical materials.
He has authored almost 100 publications on the fundamental principles and applications of plasma spectrochemical and sample preparation techniques. In addition, he has written three textbooks on trace element analysis, including a new book, which focuses on the new global directives on elemental impurities in pharmaceutical and related materials (1). He is currently editor and frequent contributor to the Atomic Perspectives Column in Spectroscopy Magazine. He has an advanced degree in Analytical Chemistry from the University of Wales in the UK and is a Fellow of the Royal Society of Chemistry (FRSC, and a Chartered Chemist (CChem).
1. Robert. J. Thomas, Measuring Elemental Impurities in Pharmaceuticals: A Practical Guide, CRC Press, Boca Raton, FL, ISBN13:978-1-138-19796 (2018)
Ion Optics
The ion optic system is usually positioned just behind or close to the skimmer cone to take advantage of the maximum number of ions entering the mass spectrometer. There are many different commercial designs and layouts, but they all have one attribute in common, and that is to transport the maximum number of analyte ions while allowing the minimum number of matrix ions through to the mass analyzer.
The ion-focusing system is not traditionally thought of as a component that needs frequent inspection, but because of its proximity to the interface region, it can accumulate minute particulates and neutral species that over time can dislodge, find their way into the mass analyzer, and affect instrument performance. Signs of a dirty or contaminated ion optic system are poor stability or a need to gradually increase lens voltages over time. For that reason, no matter what design of ion optics is used; inspection and cleaning every 3–6 months (depending on workload and sample type) should be an integral part of a preventative maintenance plan. Some useful maintenance tips for the ion optics to ensure maximum ion transmission and good stability include the following:
- Look for sensitivity loss over time, especially in complex matrices.
- If sensitivity is still low after cleaning the sample introduction system, torch, and interface cones, it could indicate that the ion lens system is becoming dirty.
- Try retuning or re-optimizing the lens voltages.
- If voltages are significantly different (usually higher than previous settings), it probably means lens components are getting dirty.
- When the lens voltages become unacceptably high, the ion lens system will probably need replacing or cleaning. Use recommended procedures outlined in the operator’s manual.
- Depending on the design of the ion optics, some single-lens systems are considered consumables and are discarded after a period of time, whereas multicomponent lens systems are usually cleaned using abrasive papers or polishing compounds, and rinsed with water and an organic solvent.
- If cleaning ion optics, make sure they are thoroughly dry because water or solvent could be sucked back into the mass spectrometer.
- Gloves are usually recommended when reinstalling an ion optic system because of the possibility of contamination.
- Do not forget to inspect or replace O-rings or seals when replacing ion optics.
- Depending on instrument workload, you should expect to see some deterioration in the performance of the ion lens system after 3–4 months of use. This is a good approximation of when it should be inspected and cleaned or replaced if necessary.
- With some instruments, you will need to break the vacuum to get to the ion optic region. Even though vacuum can be re-established very quickly, this should be a consideration when carrying out your own ion lens cleaning procedures.
Other Components
It is also important to emphasize that other components of the ICP mass spectrometer have a finite lifetime, and will need to be replaced or at least inspected and serviced from time to time. These components are not considered a part of the routine maintenance schedule, and usually require a service engineer (or at least an experienced user) to clean, service or change/replace them. These components include air filters, roughing pumps, turbomolecular pumps, mass analyzer, collision/reaction cell and ion detector.
Final Thoughts
Routine maintenance cannot be overemphasized in ICP-MS. Even though it might be considered a mundane and time-consuming chore, it can have a significant impact on the uptime of your instrument. Read the routine maintenance section of the operator’s manual and understand what is required. It is essential that time be scheduled on a weekly, monthly, and quarterly basis for preventative maintenance on your instrument. In addition, you should budget for an annual preventative maintenance contract under which the service engineer checks out all the important instrumental components and systems on a regular basis to make sure they are all working correctly.
Note: This article highlights the importance of keeping the interface and ion optic region clean, but for detailed information about maintaining the sample introduction components, please refer to Rob’s previous blog from July.
Further Reading
- Practical Guide to ICP-MS: A Tutorial for Beginners, Robert. J. Thomas; CRC Press, Boca Raton, FL, ISBN 13:978-1-4665-5543-3, June, 2014;
- Measuring Elemental Impurities in Pharmaceuticals: A Practical Guide, Robert. J. Thomas; CRC Press, Boca Raton, FL, ISBN 13:978-1-138-19796-1, February, 2018;